Periodic table chemistry 2018/26/2023 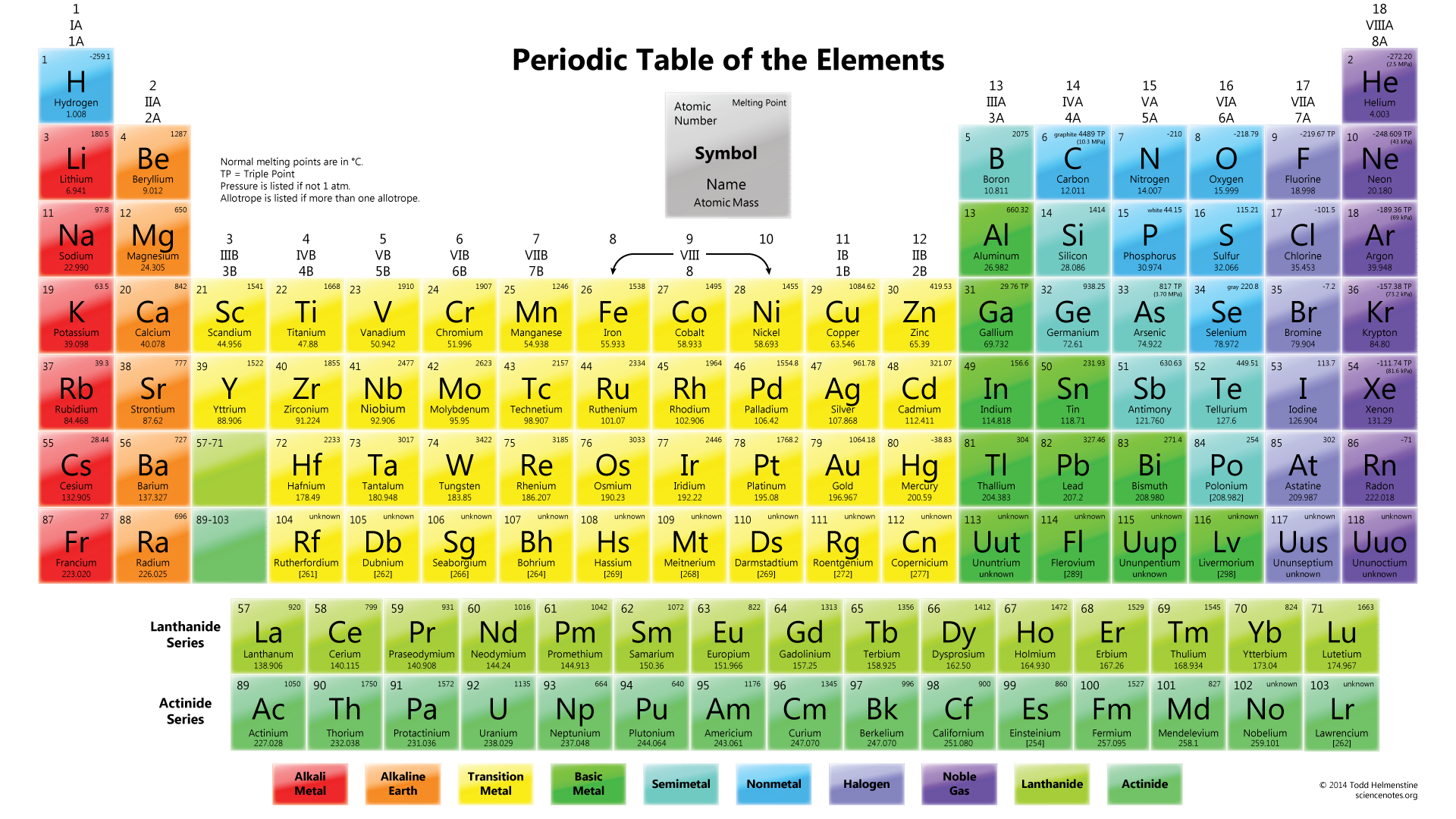 
The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium. Periodic Table - Chemistry 201 - UCalgary - StuDocu On StuDocu you find all the lecture notes, summaries and study guides you need to pass your exams with better grades. The next atom is the alkali metal lithium with an atomic number of 3. Electronic configurations and the periodic table for superheavy elements. However, we are still teaching the periodic table to Class 10 students so that they are well prepared when they get to study chemistry as a full fledged subject in Class 11, she said. The n = 1 shell is completely filled in a helium atom. All chemistry majors are advised to take General Chemistry (CHEM 111-112 or CHEM 115) and MATH 113-114 (Calculus) during the freshman year, then CHEM 201. /accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
The electron configuration and orbital diagram of helium are: For orbital diagrams, this means two arrows go in each box (representing two electrons in each orbital) and the arrows must point in opposite directions (representing paired spins). The atomic number of each element increases by one, reading from left to right. Subjects covered include: Basic atomic structure, periodic table, chemical. Period A horizontal row in the periodic table. View descriptions of chemistry courses at Loyola University Maryland. This is in accord with the Pauli exclusion principle: No two electrons in the same atom can have the same set of four quantum numbers. Members of a group typically have similar properties and electron configurations in their outer shell.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
